This perspective critically examines the hypothesis of “Mononuclear Oncogenesis” proposed by Shalin and Keskinov in their 2010 book.1 The authors proposed that malignant stem cells originate from bone marrow-derived mononuclear cells (BMMNCs) that undergo genotypic and epigenetic alterations within isolated microcavities. We compared this hypothesis with academic research published since 2010, focusing on the evolving understanding of BMMNCs and monocytes in the development of solid tumors. While early influential research, such as the 2004 study by Houghton et al.,2 suggested a direct role for BMMNCs as a source of epithelial cancers, the predominant contemporary consensus emphasizes their multifaceted contributions to the tumor microenvironment (TME), rather than their direct malignant transformation into tumor cells. This article assesses the contemporary validity of the Mononuclear Oncogenesis hypothesis, highlighting areas of convergence and divergence with current oncology research.
In 2010, Shalin and Keskinov published “Mononuclear Oncogenesis,” a theoretical treatise aimed at providing a universal and comprehensible framework for the etiology and pathogenesis of malignant tumors. This study presents a significant departure from conventional views on the origin of cancer by proposing a central role for BMMNCs in the initiation and development of solid tumors. This article delineates the core tenets of the Mononuclear Oncogenesis hypothesis and critically evaluates its standing against the backdrop of scientific advancements and research findings in oncology since its publication, particularly focusing on the period from 2010 to 2026.
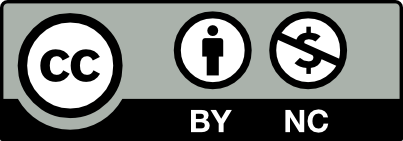
The central hypothesis of “Mononuclear Oncogenesis” posits that malignant stem cells originate from BMMNCs. The proposed mechanism is a multistage process, commencing with the formation of a “pre-tumor” bed, typically an isolated microcavity within an area of chronic inflammation. In the red bone marrow, carcinogenic stimulation is theorized to induce genotypic changes in the nuclear DNA of pluripotent or unipotent precursors of myelopoiesis. This process results in an initiated mononuclear cell (promonocyte or monocyte) that, although phenotypically normal, carries significant genotypic alterations.1
These genotype-altered mononuclear cells are believed to migrate from the bloodstream into isolated microcavities. Here, under what the authors term “super circumstances”, an aggressive, oxygen-free microenvironment, they undergo structural changes in their cell membrane and chemical alterations in their cytoplasm, leading to epigenetic modifications. The book emphasizes that mitosis serves as a crucial trigger for this transformation, during which accumulated genotypic and epigenetic alterations occur. The core mechanism of malignant transformation is described as the return of the somatic cell to an embryonic state during mitosis, a process whose differentiation is subsequently blocked owing to acquired genotypic alterations.1
This blocked differentiation, coupled with the manifestation of genotypic changes (e.g., amplifications, deletions, insertions, translocations, and micro-mutations), is proposed to lead to the gradual transformation of the undifferentiated daughter cell into a primary malignant stem cell. These emergent malignant stem cells retain the properties of the original mononuclear cell while acquiring novel capabilities, such as invasive growth, autonomy, anaerobic energy production, embryonic characteristics, uncontrolled division, and cellular immortality. The book further suggests that the potency level of the resulting malignant stem cell (pluripotent or unipotent) is contingent upon the stage at which the genotypic alterations occur in the bone marrow cell during hematopoiesis.1
Research on the role of bone marrow-derived cells (BMDCs) in cancer has undergone significant evolution since the publication of “Mononuclear Oncogenesis” in 2010. Early studies, notably the influential 2004 paper by Houghton et al.,2 provided compelling evidence that gastric cancers could originate from marrow-derived sources, suggesting the direct transformation of BMDCs into epithelial cancer cells. This finding was groundbreaking, as it challenged the prevailing dogma that tumors exclusively arise from the transformation of tissue-specific stem cells.
However, subsequent research has largely refined this understanding, shifting the primary focus from direct malignant transformation to the multifaceted contributions of BMDCs to the TME. While BMDCs, including mesenchymal stem cells and various hematopoietic cells, are recruited to tumor sites, the prevailing consensus emphasizes their role in supporting tumor growth, angiogenesis, metastasis, and immune evasion, rather than directly transforming into malignant tumor cells.3–5 BMDCs are recognized for their ability to differentiate into various stromal components, such as cancer-associated fibroblasts, pericytes, and endothelial cells, all of which are crucial for tumor progression.3,4
Monocytes, a specific type of mononuclear cell, are particularly pertinent to the hypothesis proposed by Shalin and Keskinov. Post-2010 research has extensively documented the recruitment of monocytes to tumors, where they differentiate into tumor-associated macrophages (TAMs). TAMs are critical players in the TME that influence tumor immunity, angiogenesis, and metastasis.6–8 Studies have demonstrated that cancer cells can polarize monocytes and macrophages to create a pro-tumorigenic environment, and that monocytic cells frequently constitute the most abundant immune cell population in solid tumors.7,9 Furthermore, substantial evidence indicates that monocytes undergo significant gene reprogramming in the presence of tumors.10
While the concept of BMDCs contributing to the tumor stroma is well established, direct evidence of monocytes or other BMDCs transforming into malignant epithelial cells (carcinoma) in solid tumors remains a subject of ongoing debate and is not widely accepted as a primary mechanism of oncogenesis in the post-2010 literature. The inherent plasticity of BMDCs is acknowledged; however, their malignant transformation into diverse tumor types, as suggested by earlier studies,5 has not emerged as a dominant paradigm for the origin of most solid tumors. Instead, the focus has largely shifted toward understanding their supportive, regulatory, and immunomodulatory roles within the complex TME.
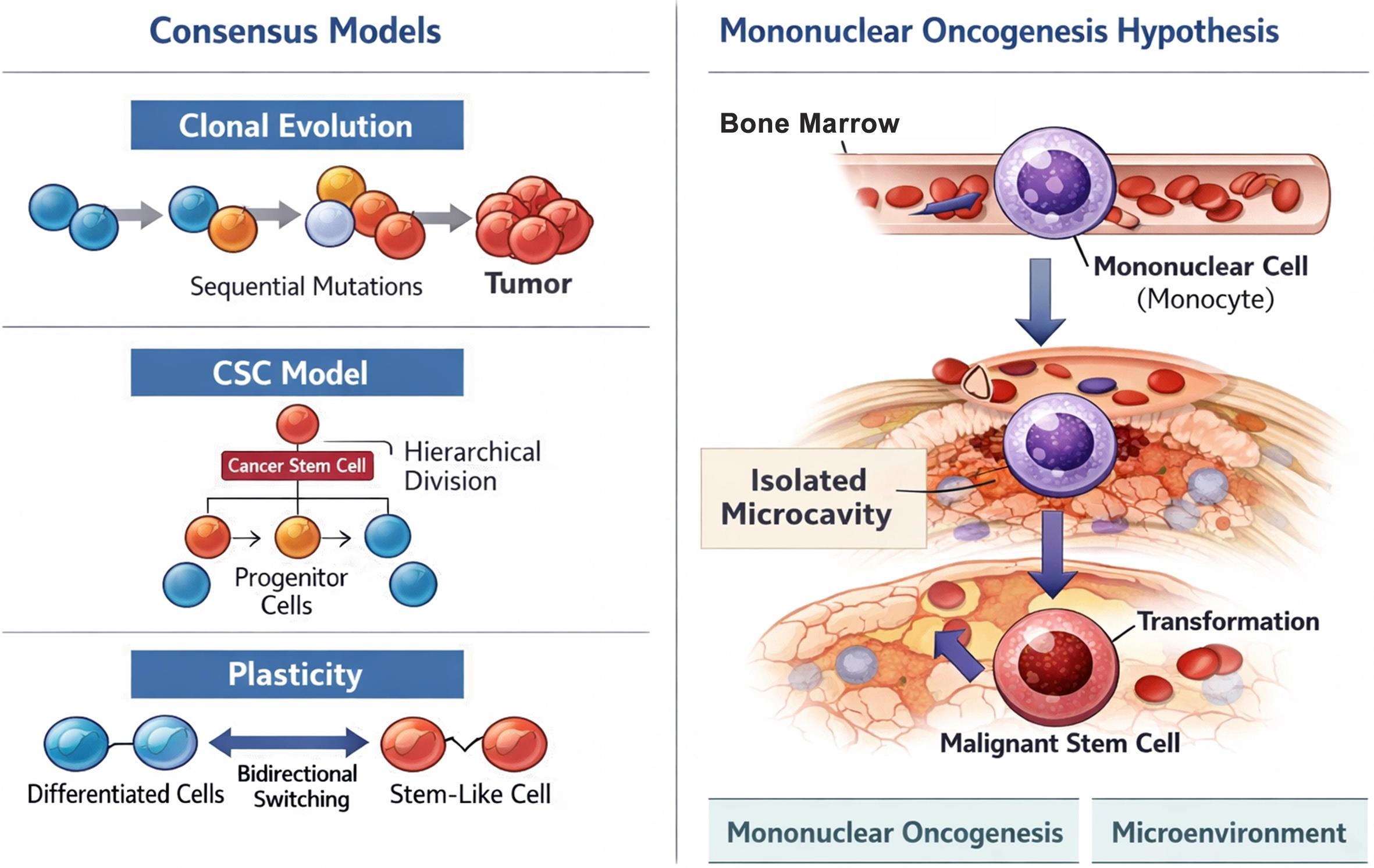
To provide a balanced perspective, it is essential to compare the Mononuclear Oncogenesis hypothesis with the three prevailing models of tumor origin: the Clonal Evolution model, Cancer Stem Cell (CSC) model, and Cellular Plasticity model (Table 1).
Key distinctions between the prevailing models of tumor origin and mononuclear oncogenesis
| Model | Primary cell of origin | Driver of heterogeneity | Role of microenvironment |
|---|---|---|---|
| Clonal Evolution | Any somatic cell | Sequential genetic mutations | Selective pressure for clones |
| Cancer Stem Cell | Tissue-resident stem/progenitor | Hierarchical differentiation | Niche support for CSCs |
| Cellular Plasticity | Dynamic (CSC ↔ non-CSC) | Epigenetic/Phenotypic shifting | Inducer of state transitions |
| Mononuclear Oncogenesis | Bone marrow mononuclear cell | Blocked differentiation of BMDCs | Primary trigger for transformation |
The Clonal Evolution model, rooted in Darwinian principles, posits that tumors arise from a single normal cell that acquires sequential genetic mutations over time. These mutations confer a selective growth advantage, leading to the expansion of the dominant clones. In this framework, any cell within a tumor has the potential to contribute to tumorigenesis if it acquires the necessary mutations. In contrast, the Mononuclear Oncogenesis hypothesis suggests a specific extratumoral origin (the bone marrow) and a specific cell type (mononuclear cells) as the primary source of the malignant lineage.
The CSC model proposes a hierarchical organization in which only a small subpopulation of cells, the CSCs, possesses the capacity for self-renewal and the ability to generate diverse cell types within a tumor. While the Mononuclear Oncogenesis hypothesis also centers on the creation of a “malignant stem cell,” it differs fundamentally in the proposed origin of these cells. The CSC model traditionally suggests that CSCs arise from the transformation of tissue-resident stem cells or progenitor cells, whereas Shalin and Keskinov argue for a systemic bone marrow-derived origin.
The Cellular Plasticity model represents a more contemporary synthesis, suggesting that cancer cells can dynamically transition between CSC and non-CSC states in response to microenvironmental cues. This model acknowledges that differentiated tumor cells can “dedifferentiate” back into a stem-like state. The Mononuclear Oncogenesis hypothesis shares some conceptual ground with this model, particularly in its emphasis on the microenvironment’s role in triggering transformation and the return of somatic cells to an embryonic state. However, the Plasticity model typically describes transitions within the tumor mass, while Mononuclear Oncogenesis describes a primary transformation event of a recruited systemic cell, as illustrated in the conceptual schematic in Figure 1.
The “Mononuclear Oncogenesis” hypothesis, while offering a novel perspective at the time of publication, presents a view of cancer origin that has been largely superseded or significantly reinterpreted by post-2010 research. The book’s central tenet, that BMMNCs directly transform into malignant stem cells that form the primary tumor, finds limited direct support in the current scientific consensus on the cellular origin of most solid tumors. While the hypothesis is bold, it remains largely speculative in its claim of a universal bone marrow origin for all solid tumors, a point that lacks robust lineage-tracing evidence the gold standard for definitively identifying the cell of origin in modern oncology required by contemporary standards.
However, it is important to acknowledge that certain elements of this hypothesis resonate with the contemporary understanding of the TME. The book’s emphasis on the role of chronic inflammation, formation of isolated microcavities, and an aggressive, oxygen-deprived environment aligns with current knowledge regarding the critical factors that promote tumor development and progression. The concepts of epigenetic changes and altered gene expression in cancer cells are well-established areas of research. Furthermore, the book’s focus on mononuclear cells (promonocytes and monocytes) as key players is partially supported by extensive research on TAMs, which are derived from monocytes and play crucial roles in tumor biology.
The primary divergence between the “Mononuclear Oncogenesis” hypothesis and contemporary oncology research lies in the ultimate fate of these mononuclear cells. Shalin and Keskinov proposed a direct malignant transformation into tumor-initiating cells. In contrast, modern oncology research, while acknowledging the remarkable plasticity of BMDCs, largely positions them as crucial components of the tumor stroma and immune infiltrates of tumors. They influence tumor behavior through complex paracrine signaling, immune modulation, and structural support rather than undergoing direct transformation into malignant cells. It is also worth noting alternative hypotheses for BMDC involvement, such as the Cell Fusion Theory, which posits that BMDCs might fuse with existing tumor cells, contributing to their plasticity and metastatic potential without undergoing direct malignant transformation.11 The idea of a somatic cell returning to an embryonic state during mitosis as a mechanism for malignant transformation, while conceptually interesting and touching upon modern concepts of dedifferentiation and CSC plasticity, is not a widely accepted or mechanistically detailed explanation in oncology. It should be noted that the evidence for such a “return to embryonic state” remains largely theoretical and lacks direct experimental validation in the context of mononuclear-to-epithelial transformation in humans.
The “Mononuclear Oncogenesis” hypothesis, as articulated in the 2010 book by Shalin and Keskinov, offers a distinctive perspective on cancer etiology, particularly through its emphasis on BMMNCs and the profound influence of the TME. While the book accurately identifies several factors now recognized as critical in cancer progression, such as chronic inflammation and the pivotal role of the microenvironment, its central claim of the direct malignant transformation of mononuclear cells into tumor-initiating cells for solid tumors is not strongly supported by the majority of post-2010 research. Current scientific understanding largely views BMDCs, including monocytes, as significant contributors to the TME, where they foster tumor growth and metastasis, but not as the direct cellular origin of malignant epithelial cells themselves. Therefore, while the book’s insights into microenvironmental influences on cancer remain relevant, its core hypothesis regarding the direct origin of malignant cells from mononuclear cells requires significant re-evaluation in light of contemporary oncology research findings.
Declarations
Acknowledgement
The author extends his appreciation to the Vice Deanship of Scientific Research Chairs, King Saud University, Saudi Arabia, for funding his research works through the Cancer Research Chair.
Funding
None.
Conflict of interest
None.
Authors’ contributions
KHF is the sole author of the manuscript.

 Author information
Author information