Introduction
IgG4-related disease (IgG4-RD) is a systemic, immune-mediated fibroinflammatory condition that has emerged over the past two decades as a unifying diagnosis for a group of disorders previously considered unrelated. It is characterized by dense lymphoplasmacytic infiltrates enriched with IgG4-positive plasma cells, storiform fibrosis, and, in many tissues, obliterative phlebitis.1–4 The disease predominantly affects middle-aged to older individuals, with a male predominance, and exhibits a chronic-relapsing course if not adequately treated.1–4
From an immunopathological standpoint, IgG4-RD is driven by a complex interplay between innate and adaptive immune responses. Aberrant activation of CD4+ T-cell subsets—particularly T follicular helper (Tfh) cells and cytotoxic CD4+ T lymphocytes—leads to B-cell differentiation, plasmablast expansion, and overproduction of IgG4 antibodies.5 Although IgG4 itself is considered a functionally monovalent, weakly proinflammatory immunoglobulin, its elevation reflects an underlying dysregulated immune milieu rather than direct antibody-mediated tissue injury. Key cytokines involved in this process include interleukin (IL)-4, IL-10, IL-21, and transforming growth factor-beta, which collectively promote class switching to IgG4, immune tolerance skewing, and progressive fibrosis.6
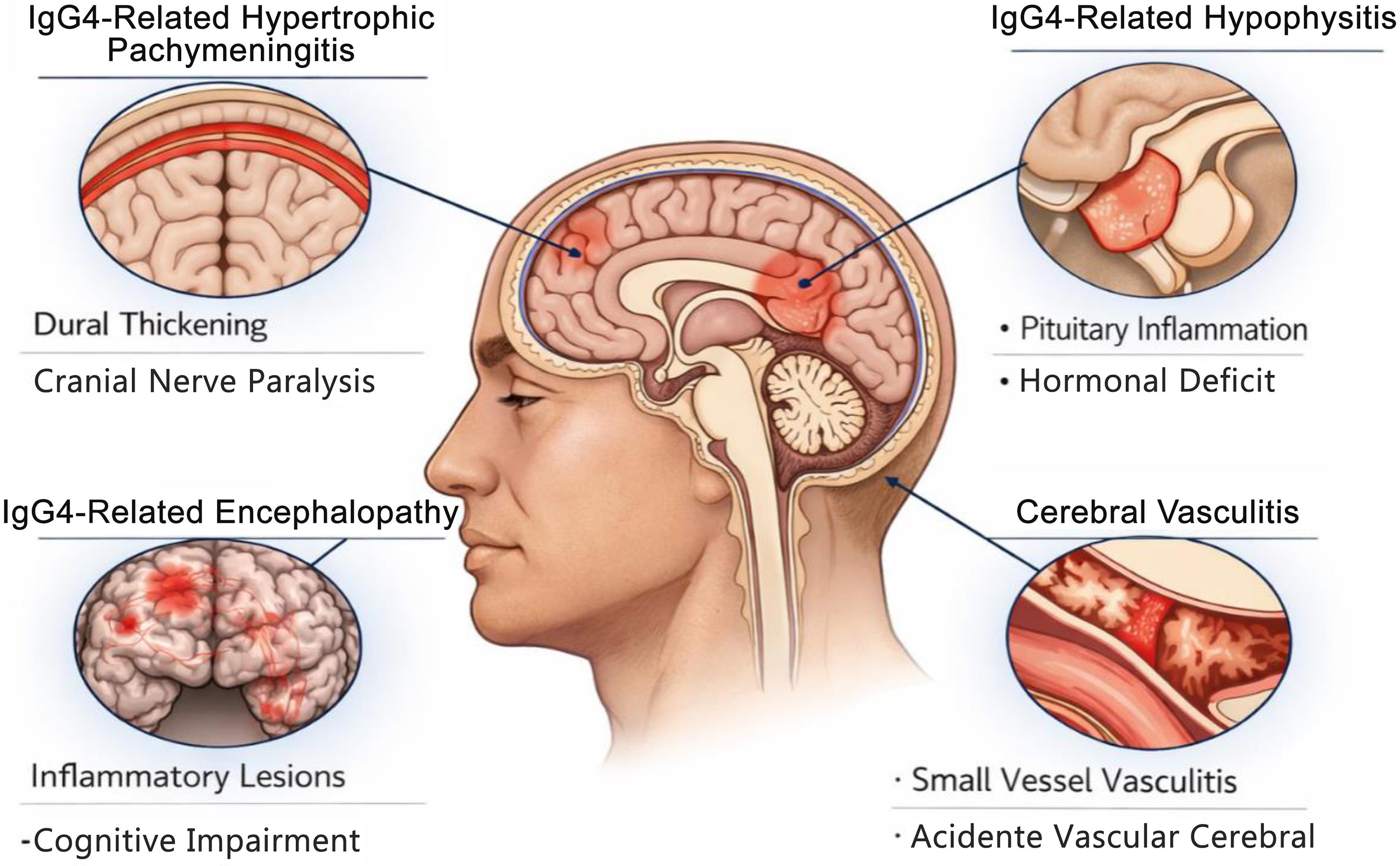
Neurological involvement in IgG4-RD, while less frequent than pancreatic, salivary, or retroperitoneal disease, is increasingly recognized as clinically significant.7 The nervous system may be affected through several converging mechanisms: direct inflammatory infiltration of neural and meningeal tissues, immune-mediated vasculopathy involving small and medium-sized vessels, and secondary ischemic or compressive phenomena resulting from fibrotic mass-like lesions.8 In the central nervous system, IgG4-related hypertrophic pachymeningitis and hypophysitis are the most characteristic manifestations, reflecting a predilection for the dura mater and sellar structures rich in connective tissue and vascular supply. These lesions demonstrate intense lymphoplasmacytic infiltration and fibrosis, leading to cranial neuropathies, headaches, endocrine dysfunction, and focal neurological deficits.9
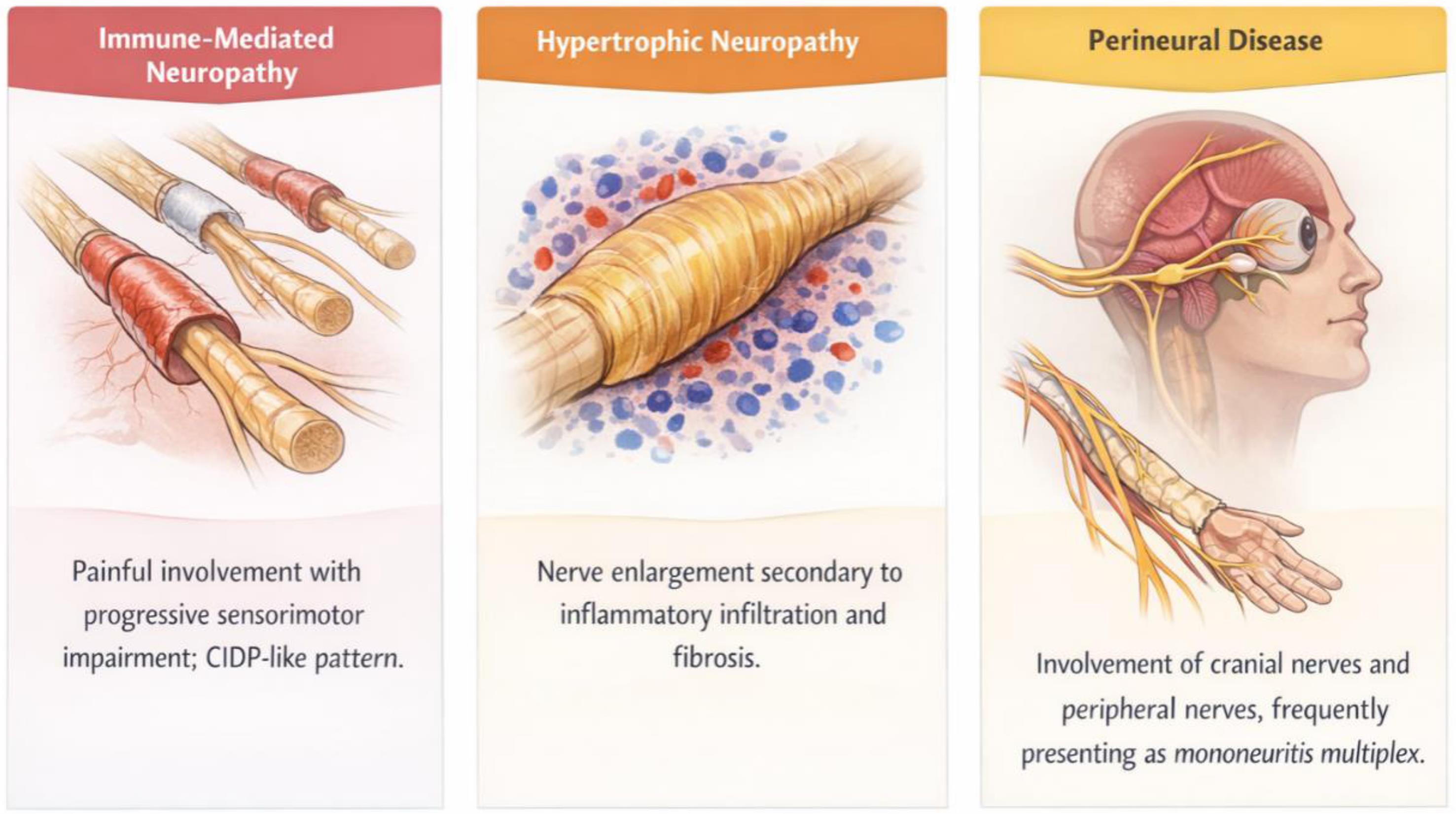
In the peripheral nervous system, IgG4-RD may present as immune-mediated neuropathy, hypertrophic nerve involvement, or perineural inflammation. Proposed mechanisms include inflammatory infiltration of the epineurium and perineurium, immune-mediated disruption of the blood–nerve barrier, and ischemic injury secondary to obliterative phlebitis of the vasa nervorum.10 Notably, some patients exhibit clinical and electrophysiological features overlapping with chronic inflammatory demyelinating polyradiculoneuropathy (CIDP), yet respond preferentially to systemic immunosuppression or B-cell–depleting therapies rather than conventional CIDP regimens.11
Because neurological manifestations may precede or overshadow systemic involvement, IgG4-RD poses a significant diagnostic challenge in neurological practice. A high index of suspicion, combined with careful assessment of systemic features, serological markers, neuroimaging patterns, and histopathological confirmation when feasible, is essential to avoid misdiagnosis and to initiate timely, disease-modifying therapy.12
This review aims to systematically summarize the clinical phenotypes, diagnostic criteria, therapeutic strategies, and recent research advances related to neurological involvement in IgG4-RD, providing evidence-based references for neurological clinical practice.
The literature was identified through searches in the PubMed/MEDLINE, Scopus, and Web of Science databases using combinations of the terms “IgG4-related disease”, “neurological involvement”, “pachymeningitis”, “hypophysitis”, and “peripheral neuropathy”. Observational studies, relevant case series, systematic reviews, and international consensus statements published in English were prioritized. Articles were selected based on clinical relevance to neurology, with emphasis on diagnostic criteria, neurological phenotypes, and therapeutic strategies.
Importance of recognizing systemic manifestations of IgG4-RD
Neurological involvement in IgG4-RD seldom occurs as an isolated phenomenon and, in most cases, reflects the systemic nature of the underlying immune-mediated process. Patients frequently present with synchronous or metachronous involvement of extraneural organs, most commonly the pancreas, lacrimal and salivary glands, kidneys, lungs, lymph nodes, and retroperitoneum.13 Recognition of these systemic features is particularly relevant in neurology, as the most frequent neurological manifestations of IgG4-RD often mimic neoplastic, infectious, or other inflammatory disorders, leading to substantial diagnostic delay.14
Among central nervous system manifestations, IgG4-related hypertrophic pachymeningitis represents the most commonly reported neurological presentation. It typically manifests with chronic or progressive headache, multiple cranial neuropathies—especially involving the optic, oculomotor, trigeminal, and facial nerves—and, in some cases, cerebellar or brainstem signs.4 Because these symptoms may precede systemic disease or appear in isolation, failure to investigate extraneural involvement can result in misdiagnosis as idiopathic pachymeningitis, meningioma, or primary central nervous system vasculitis. Similarly, IgG4-related hypophysitis is a frequent and characteristic manifestation, presenting with hypopituitarism, diabetes insipidus, and visual disturbances due to pituitary stalk and sellar infiltration.15 Recognition of associated systemic findings, such as autoimmune pancreatitis or salivary gland enlargement, is often crucial to establishing the correct diagnosis.
Peripheral nervous system involvement is increasingly recognized and constitutes another frequent neurological phenotype of IgG4-RD. Patients may present with painful, progressive sensorimotor neuropathy, mononeuritis multiplex, or hypertrophic neuropathy with nerve enlargement on imaging. In several cases, the clinical and electrophysiological presentation overlaps with CIDP. However, unlike typical CIDP, IgG4-related neuropathy often coexists with systemic fibroinflammatory disease and demonstrates a more favorable response to systemic immunosuppression or B-cell–directed therapies,11 particularly glucocorticoids and rituximab.16 Failure to recognize these systemic associations may result in suboptimal treatment and repeated relapses.
Importantly, the presence of mass-forming lesions, dural thickening, or nerve enlargement in IgG4-RD frequently leads to invasive diagnostic procedures, including neurosurgical biopsies or resections, when the systemic nature of the disease is not appreciated. Moreover, isolated neurological presentations may be erroneously classified as malignancy, granulomatous disease, or primary autoimmune vasculitis, exposing patients to unnecessary interventions or inappropriate immunosuppressive regimens.17
Given the multisystem involvement and protean neurological presentations of IgG4-RD, a multidisciplinary approach integrating neurology, rheumatology, pathology, radiology, and, when appropriate, endocrinology and gastroenterology is essential. Systematic screening for extraneural organ involvement, combined with careful neuroimaging interpretation and targeted histopathological confirmation, allows earlier diagnosis, prevents irreversible neurological damage, and facilitates timely initiation of disease-modifying therapy.18
Diagnostic criteria
The diagnosis of IgG4-RD is inherently complex and relies on the integrated assessment of clinical, serological, radiological, and histopathological findings. No single feature is pathognomonic in isolation, and this is particularly true in cases with neurological involvement, in which tissue sampling is often limited or unfeasible. Consequently, IgG4-RD is best understood as a clinicopathological diagnosis rather than a purely serological or histological entity.19
Clinical features
Clinically, IgG4-RD neurological presentations frequently reflect mass-forming or infiltrative disease processes, such as hypertrophic pachymeningitis, hypophysitis, cranial neuropathies, or peripheral nerve enlargement. Importantly, these manifestations often evolve insidiously and may lack systemic inflammatory signs, such as fever or marked elevation of acute-phase reactants, further complicating clinical recognition.14 In some patients, neurological symptoms precede systemic involvement by months or years, contributing to diagnostic delay.
Serological findings
Elevated serum IgG4 concentrations support the diagnosis but are neither sensitive nor specific. Large cohort studies have demonstrated that up to 30–40% of patients with biopsy-proven IgG4-RD have normal serum IgG4 levels, while elevated IgG4 may be observed in other conditions, including allergic diseases, infections, and malignancies.20 As such, serology should be interpreted as an adjunctive marker rather than a diagnostic criterion. Emerging biomarkers, including circulating plasmablast counts, have shown higher sensitivity for disease activity and therapeutic monitoring,21 even in patients with normal serum IgG4 concentrations.22
Imaging features
Radiological findings play a pivotal role in suggesting IgG4-RD and guiding biopsy. Typical imaging features include organ enlargement, diffuse or focal mass-like lesions, and hypertrophic inflammatory changes. In the nervous system, magnetic resonance imaging often reveals dural thickening with homogeneous contrast enhancement in hypertrophic pachymeningitis, pituitary stalk enlargement in hypophysitis, or nerve hypertrophy in peripheral neuropathy (Fig. 1). However, these findings are not specific and frequently overlap with neoplastic, infectious, or other autoimmune conditions, reinforcing the need for clinical and systemic correlation (Fig. 2).15
CNS, central nervous system; IgG4-RD, IgG4-related disease.
Histopathology (gold standard)
Histopathological examination remains the cornerstone of diagnosis and is considered the gold standard in IgG4-RD. The most widely accepted pathological features include:
Dense lymphoplasmacytic infiltrate, composed predominantly of mature plasma cells and lymphocytes;
Storiform fibrosis, reflecting a characteristic whorled pattern of collagen deposition;
Obliterative phlebitis, resulting from inflammatory destruction of venous structures;
Increased numbers of IgG4-positive plasma cells, typically defined by organ-specific thresholds and an IgG4/IgG plasma cell ratio greater than 40%.23
Notably, the full histopathological triad may not be present in all tissues or disease stages. Early or predominantly inflammatory lesions may lack storiform fibrosis, whereas advanced fibrotic lesions may show fewer IgG4-positive plasma cells. This temporal and spatial heterogeneity underscores the risk of false-negative or indeterminate biopsy results, particularly when small or non-representative samples are obtained.
Diagnostic challenges and the role of clinicopathological correlation
A major diagnostic challenge in IgG4-RD is the potential dissociation between clinical manifestations and histopathological findings. In neurological disease, direct biopsy of affected tissue is often high risk or technically unfeasible, and histological confirmation must rely on extraneural tissue.24 Furthermore, some patients exhibit typical clinical and radiological features of IgG4-RD despite incomplete or atypical histopathological findings, while others meet pathological criteria in the absence of overt neurological symptoms. For these reasons, current international consensus emphasizes the importance of clinicopathological correlation, integrating clinical phenotype, imaging patterns, serological data, and histology from the most accessible affected organ.25 Multidisciplinary case discussion is strongly recommended to avoid both overdiagnosis and underdiagnosis, which may delay effective immunomodulatory therapy and result in irreversible neurological damage.
The analysis presented in Table 1 consistently demonstrates that neurological involvement in IgG4-RD is not an exceptional occurrence but rather an integral component of a well-defined systemic spectrum with reproducible clinical patterns.4,7,14 Multicenter cohorts and systematic reviews consistently support the notion that hypertrophic pachymeningitis and hypophysitis represent the predominant central nervous system phenotypes in IgG4-RD,26 commonly associated with synchronous or metachronous extraneural disease. These studies also report high rates of initial response to systemic glucocorticoid therapy, reinforcing the immune-mediated inflammatory nature of the condition.27 Nevertheless, longitudinal data indicate a substantial relapse risk in patients who do not receive maintenance immunosuppression, underscoring the need for long-term therapeutic planning in neurological practice.28
Major studies reporting neurological manifestations of IgG4-RD
| Author/Year | Study type | N (patients) | Neurological manifestations | Key findings | Limitations |
|---|---|---|---|---|---|
| Khosroshahi et al., 20157 | Multicenter cohort | 125 | Pachymeningitis, hypophysitis | CNS involvement frequently associated with multisystem disease | Retrospective design with limited long-term neurological follow-up |
| Lu et al., 20144 | Systematic review | 87 | Predominant hypertrophic pachymeningitis | High response rates to corticosteroid therapy | Heterogeneity of included studies and lack of standardized outcome measures |
| Wallace et al., 201914 | Observational cohort | 60 | Cranial neuropathies, CNS involvement | Relapses common in the absence of maintenance therapy | Limited sample size and variability in treatment strategies |
An additional and clinically relevant insight emerging from the studies summarized in the table is the heterogeneity of neurological phenotypes and their frequent overlap with other inflammatory or neoplastic disorders. Several reports emphasize that IgG4-RD may mimic primary central nervous system vasculitis, meningioma, neurosarcoidosis, or idiopathic hypertrophic pachymeningitis, whereas peripheral nervous system involvement is often misdiagnosed as CIDP or nonspecific immune-mediated neuropathy. This phenotypic overlap contributes to diagnostic delay and underscores the importance of recognizing systemic disease patterns, characteristic imaging findings, and supportive histopathological features.29 The literature further demonstrates that neurological manifestations are frequently the presenting feature prompting medical evaluation, reinforcing the neurologist’s pivotal role in initiating appropriate diagnostic pathways.
From an evidence-based clinical perspective, the accumulated data strongly support systematic diagnostic suspicion and early integration of multidisciplinary evaluation when encountering potentially treatable inflammatory neurological syndromes. Recent advances in the understanding of IgG4-RD pathogenesis and treatment outcomes have demonstrated that accurate diagnosis enables effective disease control with immunosuppressive and B-cell–directed therapies, resulting in neurological stabilization or improvement in a substantial proportion of patients.30 Importantly, delayed recognition is associated with irreversible fibrotic changes and permanent neurological deficits. Therefore, the findings summarized in Table 1 provide a robust framework to support daily clinical decision-making, emphasizing that IgG4-RD should be routinely considered in the differential diagnosis of unexplained inflammatory neurological disorders and managed according to evidence-based strategies.
The data summarized in Table 2 clearly demonstrate that IgG4-RD,7,15,21,24,25,31 including its neurological manifestations, is a highly treatment-responsive condition when therapy is initiated at an early inflammatory stage. Across multiple cohorts and systematic reviews, glucocorticoids remain the cornerstone of induction therapy, achieving rapid clinical and radiological improvement in the majority of patients.32 However, the table also highlights a consistently high relapse rate when corticosteroids are used as monotherapy, particularly in patients with central or peripheral nervous system involvement.33 As previously discussed, neurological involvement in IgG4-RD typically shows an initial response to glucocorticoids, but relapse is common without maintenance therapy.
Therapies for IgG4-related disease with neurological involvement
| Therapy | Mechanism | Evidence | Efficacy | Safety profile | Neurological evidence | Limitations |
|---|---|---|---|---|---|---|
| Glucocorticoids7,15 | Broad immunosuppression | First-line | High initial response | Relapse, metabolic effects | Effective for CNS and PNS manifestations | High relapse rates and long-term toxicity |
| Azathioprine7 | Antimetabolite | Maintenance | Moderate | Myelotoxicity | Used mainly as steroid-sparing agent | Limited neurological-specific data |
| Mycophenolate mofetil7 | Lymphocyte inhibition | Maintenance | Moderate | Gastrointestinal intolerance | Adjunctive therapy in refractory cases | Evidence based on small cohorts |
| Methotrexate7 | Antimetabolite | Adjunct | Variable | Hepatotoxicity | Occasional use in neurological IgG4-RD | Inconsistent efficacy and limited studies |
| Rituximab21 | Anti-CD20 B-cell depletion | Strong evidence | High, relapse reduction | Infusion reactions | Effective in refractory CNS and PNS disease | Relapses may occur after B-cell reconstitution |
| Inebilizumab24,25,31 | Anti-CD19 B-cell depletion | Emerging | Promising | Long-term safety under evaluation | Reported benefit in refractory neurological IgG4-RD | Limited long-term and real-world data |
This finding reinforces the concept that delayed diagnosis or insufficient maintenance therapy allows progression toward irreversible fibrosis, which is associated with poorer neurological outcomes. Consequently, early recognition and prompt initiation of immunomodulatory treatment are critical to preserve neurological function and prevent permanent disability.
Importantly, Table 2 underscores the growing role of targeted B-cell therapies, reflecting advances in the understanding of IgG4-RD pathophysiology. While anti-CD20 agents such as rituximab have demonstrated robust efficacy by depleting mature B cells and reducing plasmablast populations, emerging evidence suggests that broader B-cell–directed therapies may provide deeper and more sustained immunological control in relapsing or neurologically aggressive IgG4-RD phenotypes.34
Use of inebilizumab (anti-CD19) in IgG4-RD — current therapeutic positioning
Inebilizumab is a humanized monoclonal antibody targeting CD19, expressed across a broader spectrum of the B-cell lineage than CD20, including naive and memory B cells, plasmablasts, and certain plasma cell precursors. By depleting CD19+ populations, inebilizumab provides a mechanistically plausible advantage in IgG4-RD, given the pathogenic expansion of plasmablasts and oligoclonal CD4+ T-cell responses observed in active disease.35 Recent multicenter data have demonstrated clinically meaningful responses in multiple organ systems, including cases with neurological involvement, with reductions in disease activity scores, improvement in organ function, and significant steroid-sparing effects.31 Inebilizumab was approved by the U.S. Food and Drug Administration for the treatment of IgG4-RD in April 2025, based on the results of a phase 3 clinical trial published in late 2024. In clinical practice, inebilizumab currently represents a compelling option for patients with relapsing neurological involvement in IgG4-RD, those with contraindications to long-term corticosteroid therapy, or cases refractory to rituximab.36,37
Future directions and ongoing research
One of the major unmet needs in IgG4-RD is the identification of reliable biomarkers capable of reflecting disease activity and predicting relapse, particularly in patients with neurological involvement. Serum IgG4 concentrations lack sufficient sensitivity and specificity, and their levels often do not correlate with clinical activity.38 Recent studies have demonstrated that circulating plasmablast counts correlate more closely with disease burden and treatment response,39 even in patients with normal serum IgG4 levels. Additional candidate biomarkers under investigation include Tfh cell subsets, cytokine signatures, and soluble IL-2 receptor levels.40 The validation of such biomarkers may significantly enhance longitudinal monitoring and guide individualized therapeutic strategies in neurological involvement in IgG4-RD.
Optimal duration of B-cell–depleting therapy
Although B-cell–depleting therapies have transformed the management of IgG4-RD, the optimal duration and retreatment strategy remain undefined. Observational studies suggest that fixed-duration therapy may be insufficient for patients with severe or relapsing neurological disease, while prolonged B-cell depletion raises concerns regarding infection risk and immune reconstitution dynamics.41 Current research efforts are focused on defining relapse predictors, immune reconstitution patterns, and biomarker-guided retreatment protocols, particularly relevant for neurological phenotypes in which disease recurrence may result in irreversible structural damage and long-term disability.
Emerging biologics targeting the B-cell lineage and plasmablast compartment (inebilizumab and beyond)
Advances in the understanding of IgG4-RD immunopathogenesis have strengthened the rationale for therapies targeting earlier and broader checkpoints of B-cell differentiation. Inebilizumab, by targeting CD19, affects naive and memory B cells, plasmablasts, and a fraction of IgG4-producing plasma cell precursors, thereby modulating the cellular populations most closely associated with disease activity and relapse.35 Ongoing translational studies are evaluating whether CD19-directed therapy offers superior relapse prevention compared with anti-CD20 therapy, particularly in patients with neurological involvement or multiorgan disease.24 In parallel, novel biologics targeting plasma cell survival pathways and Tfh–B cell interactions, including BAFF and APRIL inhibitors, are under investigation and may complement or supersede current B-cell–depleting strategies.42 These therapeutic developments highlight a broader paradigm shift in IgG4-RD toward mechanism-based immunotherapy, with particular relevance to central and peripheral nervous system involvement, where irreversible fibrosis remains a major determinant of functional disability. Despite these promising results, long-term data on neurological outcomes, relapse prevention, and sustained safety of anti-CD19 therapy in IgG4-RD remain limited, particularly for central nervous system involvement, warranting cautious interpretation and continued longitudinal studies.
Long-term neurological outcomes and neuroimaging correlates
Long-term outcome data in neurological involvement in IgG4-RD remain limited, particularly regarding cognitive outcomes, functional recovery, and neuroimaging evolution. Emerging longitudinal MRI-based assessments, including volumetric and quantitative analyses, may offer tools to evaluate treatment response and residual structural damage in hypertrophic pachymeningitis and hypophysitis.43 These efforts are essential for refining prognostic models and optimizing long-term management strategies.
Conclusions
IgG4-RD represents an important, frequently underrecognized, and potentially reversible cause of neurological disease. Awareness of its diverse central and peripheral nervous system manifestations is critical for timely diagnosis and appropriate treatment. The growing body of evidence supports early initiation of immunomodulatory therapy, guided by clinicoradiological and pathological correlation, to prevent irreversible neurological damage. Advances in targeted immunotherapy—particularly B-cell–directed and plasma cell–oriented agents—are reshaping the therapeutic landscape and offering improved long-term neurological outcomes for patients with neurological involvement in IgG4-RD. Continued research into biomarkers, optimized retreatment strategies, and translational immunology will further enhance evidence-based care.
Declarations
Acknowledgement
None.
Funding
None.
Conflict of interest
The author declares no competing interests.
Authors’ contributions
FTMO is the sole author of the manuscript.

 Author information
Author information